After a decade in the pharmaceutical industry where she had a hand in developing prominent drugs including the immunosuppressive drug Humira, Shao-Lee Lin started her own biopharma company.
She founded Acelyrin in 2020, armed with $8 million in Series A funding. It was one of 165 Series A funding rounds in the space that year. In 2021, Lin said Acelyrin had the potential to become “the next Amgen,” a pharma company that has been around since the ’80s.
“We really are after building a long-term sustainable biopharma company that ultimately has fully integrated R&D as well as commercialization,” Lin told me when I was a reporter at dot.LA.
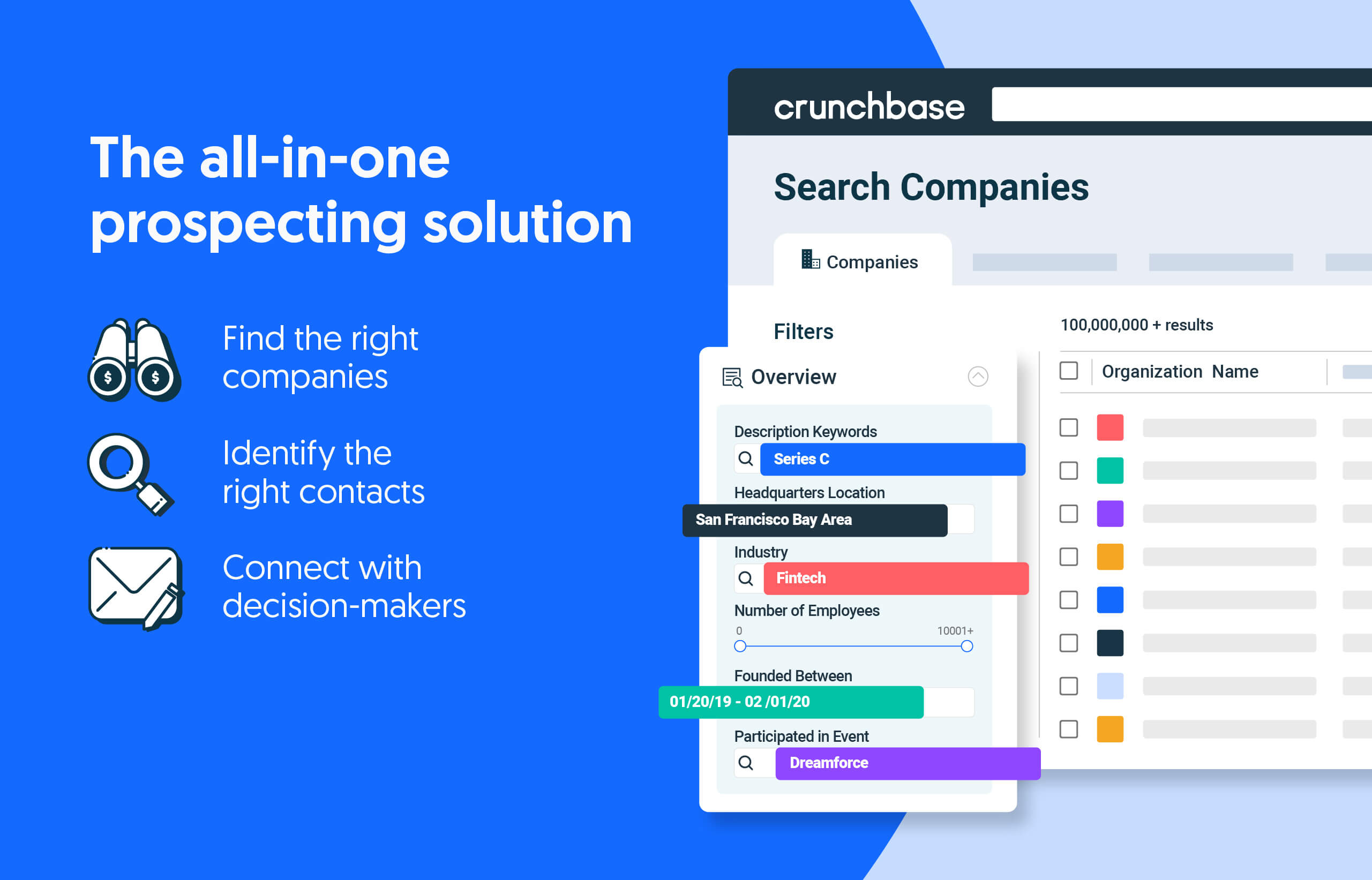
Search less. Close more.
Grow your revenue with all-in-one prospecting solutions powered by the leader in private-company data.
That reality is rare for up-and-coming biotechs. Of the top six pharmaceutical companies in the world ranked by 2021 revenue, four were founded in the 1800s. They’re names you know well: Johnson & Johnson, Merck, Pfizer and Roche.
But it has often been difficult for new entrants to join their ranks. More than $664 million in seed and pre-seed venture funding flowed to biotech startups in 2022, with more than $90 million raised so far in 2023. Yet none of these millions are likely to help build the next large pharma company on the scale of Merck and Pfizer. The steep costs of R&D and commercialization, as well as VC expectations for returns on their investments, are too complex for these often one-molecule startups, which can’t scale fast enough to compete.
“You rarely, these days, find aspirations of becoming a fully integrated pharmaceutical company,” said David Crean, a biotech investor who sits on the board of startups including Histogen and Amydis. “You very rarely will find somebody who says, ‘Hey, I want to be the next Amgen.’ ”
In 2022, pharma seeded 228 rounds that amounted to more than $664 million in funding, per Crunchbase data. History tells us that almost all of the funded startups will ultimately sell to a big pharma company.
An expensive venture
First, a quick primer on the typical lifecycle of a drug startup: Research begins at the university level, powered by grants. If a group of scientists finds a promising molecule that could develop into a drug, they will start a company. That startup is now on the hunt for funding that will power further research and development, as well as preclinical and clinical trials. The trials are meant to prove the drug is safe and efficacious, which is when the startup will usually try to sell to a bigger company.
“The reason they target that time to sell is because [trials] are expensive, but relatively inexpensive [compared to the rest of the process],” said Todd Thomson, COO and CFO of Kairos Ventures. “And now you’ve proven you’ve taken some of the big risks away and you can get a decent price.”
The later trials are often far more expensive and risky — the entire drugmaking process takes around 10 years and costs billions of dollars. In the process, 90% of drugs will fail.
The commercialization issue
Perhaps the biggest differentiator between small biotech startups and the huge pharma companies are in commercialization. Startups generally direct all their funding toward R&D in the lab, tinkering away at one or two promising molecules. But large pharma companies like Amgen have entire departments working on marketing strategies, intellectual property management, and sales pitches for several drugs. That is why so many startups are discouraged from forming commercialization departments.
“As an adviser, I tell startups all the time ‘don’t pursue it, it’s very, very expensive,’ ” Crean said. “To build a commercial infrastructure is incredibly expensive, and I think stupid, in terms of utilizing your capital.”
A good example of the intense commercialization process is Novo Nordisk and its push to commercialize the extremely popular weight-loss drug Ozempic, which was originally designed to lower blood sugar. The company sent fleets of pharmaceutical sales representatives knocking on doctors’ doors. It began a marketing campaign called “It’s Bigger Than Me” and nabbed Queen Latifah as a spokesperson. It even funded new coursework materials around obesity that would eventually be taught in medical schools.
Funding that kind of endeavor is near impossible for most startups, which usually have one or two molecules to their name.
“The only thing you have is that one other molecule, and now you have to stand up a sales force to try to sell this to all these doctors and hospitals across the U.S. and then across the world?” Thomson said. “It’s a very expensive proposition.”
New players
Acelyrin isn’t the only company trying to become a fully realized pharmaceutical company complete with R&D and commercialization departments. Kairos Ventures, which primarily invests in seed-stage biotech startups, has seen several founders of nascent companies who had hopes of competing in the big leagues.
“I think if you ask them directly what they really feel, they want to become the next Pfizer. That’s their ambition,” Thomson said. “If they really are going to go in that direction, then they’re going to need a lot more capital that we can provide.”
Combined, Johnson & Johnson, Merck, Pfizer and Roche netted a healthy $2.9 trillion in revenue in 2021. That explains why new entrants have ambitions to get to the top.
And some new investors are looking to support exactly that. Time BioVentures is a new venture firm started in 2022 by biotech veteran Timothy Wright and rock star D.A. Wallach.
“The investment community [in biotech] tends to be very ‘asset centric.’ So when they look at a company that’s developing a portfolio of drugs, they’re thinking about it in terms of what each asset is worth and how likely each asset is something that a big pharmaceutical company would want to come and buy,” Wallach said. “They sometimes miss the broader picture, which is that an entrepreneur is trying to build a long-lasting company.
“A company is like a living organism, right?” Wallach added. “It’s a group of people who bring their talents together and bring capital together to do something innovative for a long time.”
But investing in lasting companies, especially in a sector as slow-moving as biotech, means investors are less likely to see a return on investment anytime soon. The current model of innovate-and-exit isn’t bound to phase itself out anytime soon.
“It’s a quick return to the market, to give money back to the shareholders on their growth in the equity,” Crean said. “And that, to me, is a more efficient model.”
Illustration: Dom Guzman

Stay up to date with recent funding rounds, acquisitions, and more with the Crunchbase Daily.




![M&A - Illustration of a magnet attracting various products. [Dom Guzman]](https://news.crunchbase.com/wp-content/uploads/mergers_and_acquisitions-470x352.jpg)
![Cybersecurity illustration with shield, books, laptop. [Dom Guzman]](https://news.crunchbase.com/wp-content/uploads/Cybersecurity_Shield-470x352.jpg)
![Illustration of a guy watering plants with a blocked hose - Global [Dom Guzman]](https://news.crunchbase.com/wp-content/uploads/quarterly-global-3-300x168.jpg)
67.1K Followers