By Michael May
Canada is clean and boring, with a publicly-funded health system. Although we Canadians tend to roll our eyes at that characterization, it contains a certain amount of truth. In fact, for those of us who work in biotech—especially cell and gene therapy manufacturing—it’s exactly the point.
Cell and gene therapy (CGT) is the frontier of modern medicine. Unlike conventional drugs, these living therapies are specifically engineered to directly attack diseases and cure them, instead of just treating the symptoms. Around the world, researchers and startup companies are working on revolutionary new treatments for cancer, arthritis, diabetes, liver disease, Alzheimer’s and more.
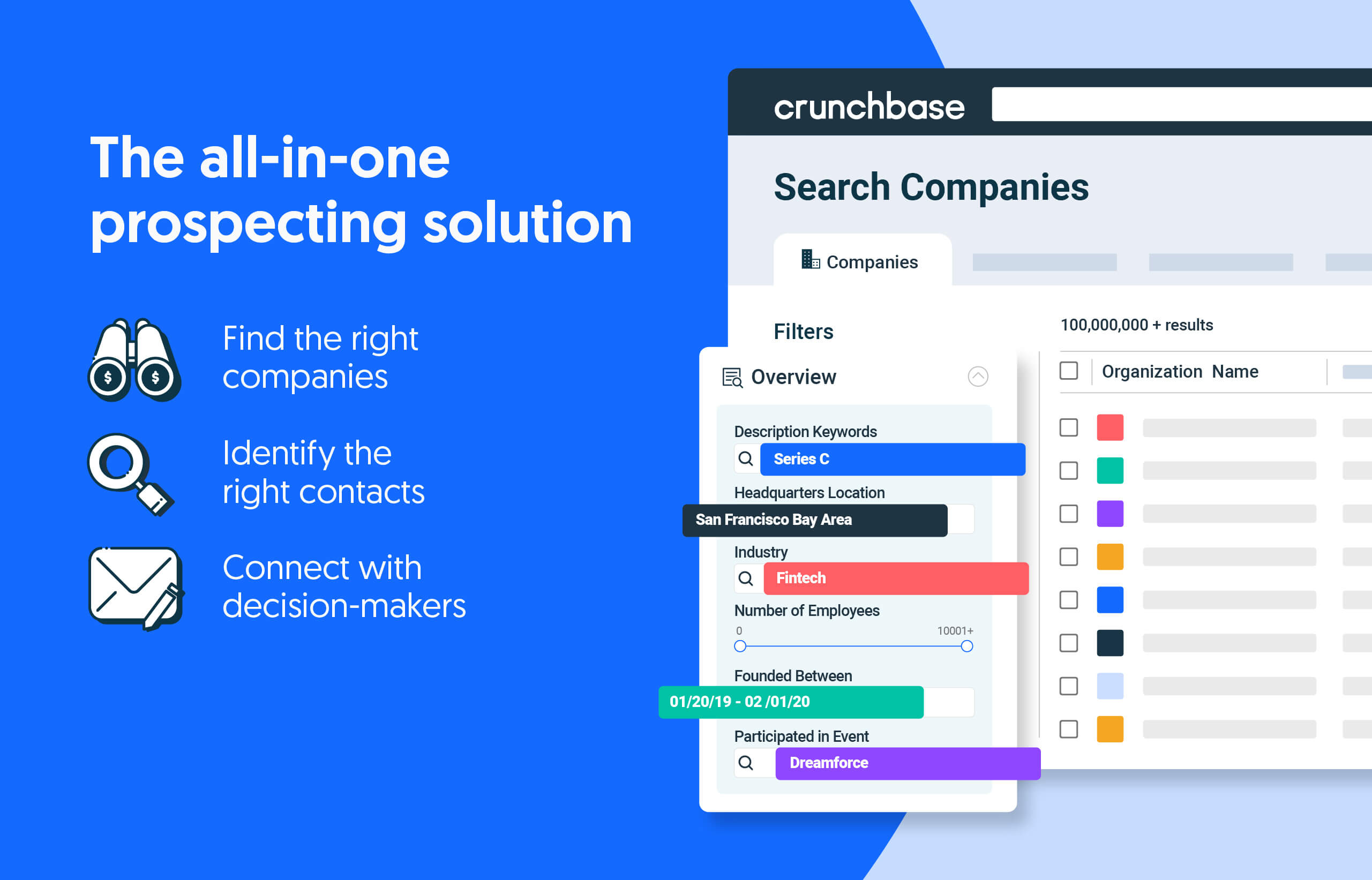
Search less. Close more.
Grow your revenue with all-in-one prospecting solutions powered by the leader in private-company data.
But like so many others, the biotech industry has been deeply challenged by COVID-19.
We all know about the Herculean efforts by vaccine makers, but CGT manufacturing has also been severely disrupted by the pandemic. Cell donors have been isolated, borders have been closed, flights have been curtailed or delayed. Components and materials have been caught up in broken supply chains. Biotech talent shortages and remote-work protocols have kept CGT manufacturers working at reduced capacity.
What’s a CDMO?
These are huge obstacles, because manufacturing is critical for CGT trials, approvals and product commercialization. And because building more manufacturing capacity is a costly business. Some of the biggest players have the hundreds of millions of dollars required to build their own facilities, but most firms simply can’t afford that, especially emerging companies.

These small, innovative ventures need contract development and manufacturing organizations (CDMOs) to help scale their revolutionary research into viable commercial treatments.
CDMOs are a growing and critical link in what’s expected to become a $34 billion annual market for CGTs by 2030. Canada is already a player in this burgeoning industry, thanks in part to its excellent publicly funded universities and health system. Two University of Toronto researchers discovered stem cells back in the 1960s, and the same network continues to churn out talent, research and startups today.
Commercialization
But, until recently, Canadians weren’t doing all we could to commercialize this work. Many of our scientists and startup companies ended up taking their work to the United States, or farther afield to hubs in Europe and Asia. But that’s beginning to change, thanks to targeted public investment and a greater understanding of the importance of CGT manufacturing.
A solid example of an organization advancing CGT manufacturing commercialization in Canada is Toronto-based CCRM—which also happens to be where I serve as President and CEO. In addition to seeding and nurturing a dozen promising CGT startups, we’re breaking ground on OmniaBio, a 250,000 square foot (initially) biomanufacturing plant at McMaster Innovation Park in Hamilton, Ontario, just an hour from Buffalo.
Underwritten by public funding from the government of Ontario, which is helping to build up the province as a biomanufacturing hub, OmniaBio’s clean rooms, lab space and targeted services will instantly become Canada’s largest and most advanced CDMO for CGTs, a key cog in the country’s growing ecosystem.
But this facility will also add desperately needed contract manufacturing capacity to the broader North American market, which is significantly bottlenecked. The number of therapies in development are outpacing the amount of space and talent required – the U.S. Food and Drug Administration expects to be approving as many as 20 new cell and gene therapies a year by 2025. CDMOs that can help biotech companies through the development process and facilitate manufacturing at scale will be key to getting these therapies commercialized.
Canada might not be top-of-mind for some U.S. ventures, but a healthy number of American companies have already shown a willingness to do this work in Canada. We’ve seen them at our existing facilities at MaRS Discovery District in Toronto, and we’ve seen them in the pipeline of customers lining up to work at the new facility in Hamilton. For innovators with precise standards, boring can be beautiful if it means a pristine facility subsidized by public investment, just 50 miles from the border.
Dr. Michael May is the president and CEO of CCRM. Previously, he was the president and co-founder of Rimon Therapeutics Ltd., a Toronto-based tissue engineering company developing novel medical polymers that possess drug-like activity.
Illustration: Li-Anne Dias.
Stay up to date with recent funding rounds, acquisitions, and more with the Crunchbase Daily.



![Illustration of a guy watering plants with a blocked hose - Global [Dom Guzman]](https://news.crunchbase.com/wp-content/uploads/quarterly-global-3-300x168.jpg)
67.1K Followers