Clinical trials are the make-or-break step to financial success for a drug that took decades to make. Without them, it can’t get approved by the Food and Drug Administration.
Trials have always been challenging to execute, but now the makeover that began during the pandemic is getting a refresh.
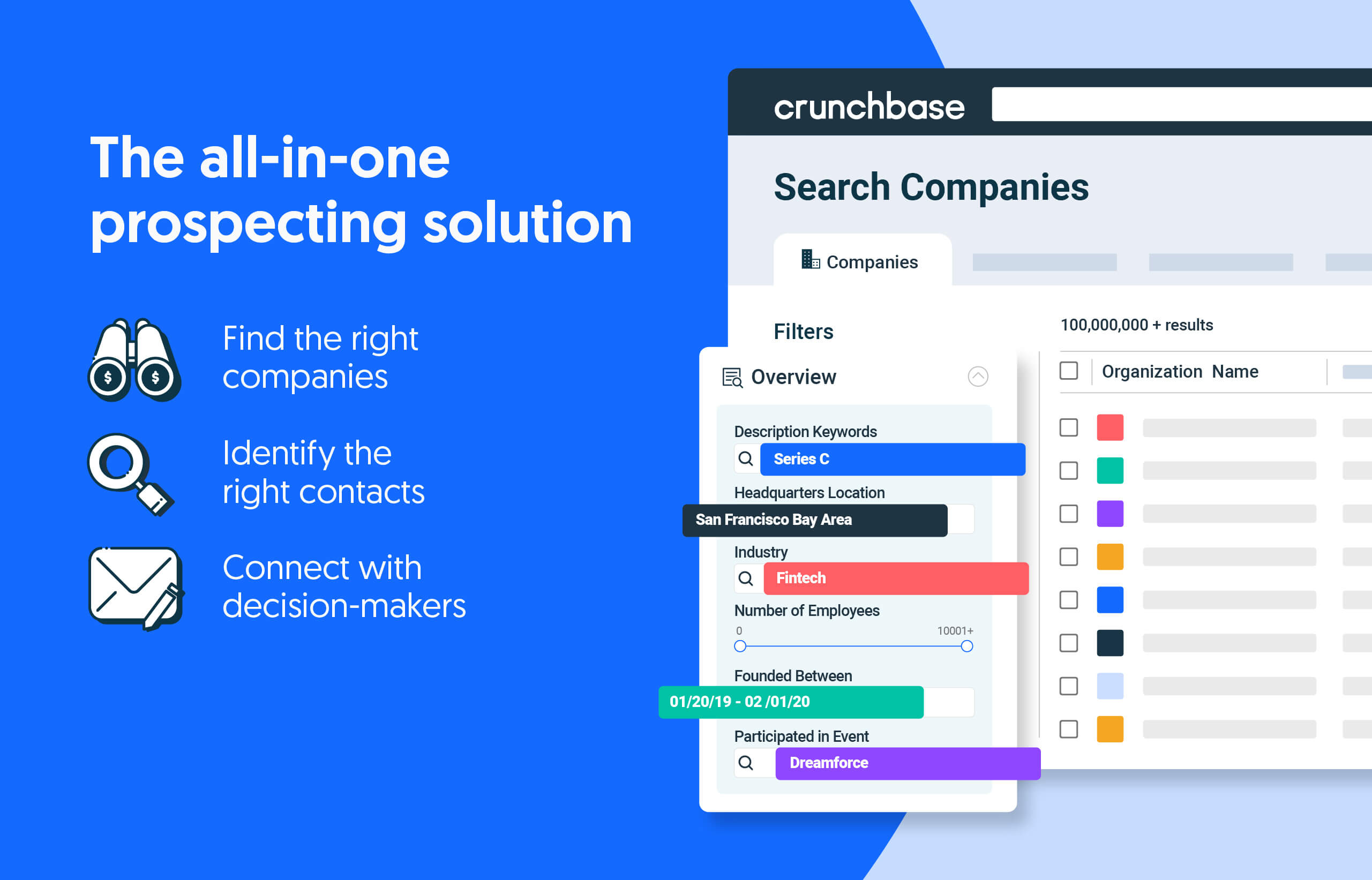
Search less. Close more.
Grow your revenue with all-in-one prospecting solutions powered by the leader in private-company data.
Enter Vial, a San Francisco-based startup designed to organize the trial process. The company announced on Wednesday it raised $67 million in funding led by General Catalyst with additional participation from the likes of Byers Capital and BoxGroup.
Clinical trials have undergone massive changes since the pandemic when the FDA outlined new policies allowing them to continue in a remote environment. As a result, a slew of startups like Vial have entered the clinical trial space to make the process faster and more efficient while troubleshooting problems that may result in the FDA not approving a drug.
What’s the problem?
Vial is tackling one of the most tedious problems in biotech: slow-moving and capital-intensive clinical trials. The clinical trial process, by which drugs are tested on humans, requires proper execution in order to be approved by the FDA.
But there are a lot of inefficiencies in the process: companies need to cherry-pick the right candidates for their drug, and there are often age or disease-severity requirements associated with being qualified to take the drug. Those candidates also need to be able to visit a health office frequently for clinicians to collect a slew of biometric and behavioral data from them throughout the clinical trial.
Vial’s platform allows companies to find patients across the country and better collect data that can be organized. Through its suite of applications, research organizations can manage patients worldwide on one platform, have a central hub for communicating and organizing participants and collect data in a uniform manner.
Companies like Vial have seen massive funding since the pandemic. In 2021 funding peaked at over $2 billion, according to Crunchbase data.
A new profit-making venture
Big box stores are getting involved too. In October, Walmart announced it would jump on the clinical trial train by identifying patients that may qualify as test subjects for a certain drug or product.
Through its pharmacy patient portal, patients get recommendations for opportunities they can participate in. In June, Walgreens launched its own clinical trial department and in February, CVS partnered with Medable to provide clinical trial services at its pharmacy-clinics.
Several of these companies bill their services as a way to get more diversity into clinical trials. Indeed, a decentralized clinical trial process is easier to onboard patients without asking them to commit to a full schedule of visiting testing sites for years, opening the door for people with disabilities, lower income individuals and people of color to participate in testing drugs that would enter the market.
In reality, pharmacy data from big companies like Walmart and CVS are likely huge untapped streams of revenue from biotech companies willing to spend to find the right participants for their clinical trials.
Biotech companies are racing against their drug patents, which expire after around 20 years to make way for generics. That means every inefficiency in the drug approval process can cost a company extra years of would-be profit.
Illustration: Dom Guzman

Stay up to date with recent funding rounds, acquisitions, and more with the Crunchbase Daily.



![Illustration of agentive AI brain - North America - Quarterly. [Dom Guzman]](https://news.crunchbase.com/wp-content/uploads/Agentive_AI_n-america-470x352.jpg)
![Illustration of a guy watering plants with a blocked hose - Global [Dom Guzman]](https://news.crunchbase.com/wp-content/uploads/quarterly-global-3-300x168.jpg)
67.1K Followers